Editor’s note: This story contains a graphic image of animal mistreatment.
The United States Department of Agriculture issued a “critical” violation to VUMC during its April 27, 2021, annual inspection. This violation came as a result of guinea pig mistreatment during an Aug. 2020 study involving intubation and ventilator testing.
The Investigative and Enforcement Services department of the USDA officially warned VUMC concerning the same incident on June 4, 2021.
“If APHIS [Animal and Plant Health Inspection Service agency of the USDA] obtains evidence of any future violation of these federal regulations, APHIS may pursue civil penalties, criminal prosecution, or other sanctions for this alleged violation(s) and for any future violation(s),” the warning read.
The incident was initially self-reported by VUMC to the NIH’s Office of Laboratory Animal Welfare and formalized by the April 2020 USDA inspection report. The report also stated that VUMC “corrected” the issue before its annual inspection.
“All incidents appearing in USDA inspection reports are self-reported by VUMC to regulatory officials and we work closely with these officials on any process improvements that may be needed,” an official VUMC statement sent to The Hustler said.
Lyndsay Cole, APHIS assistant director of public affairs, stated that there can be delays between when a violation is issued and when the incident occurred. She said facilities work with their Institutional Animal Care and Use Committee, which enforces the Animal Welfare Act, to promptly record and correct the circumstance, which is then “formalized” on the USDA inspection report.
“Depending on the situation, facilities are not required to immediately report the incident to an animal care inspector if it has been corrected [by their IACUC],” Cole said. “If there’s something that is ongoing and a direct threat to the animals, we will address it right away.”
Cole also stated that 97 percent of all USDA-licensed facilities—including breeders, dealers, exhibitors, research labs and transporters—are in compliance with the AWA.
April 2021 citation
According to the 2021 inspection report, four guinea pigs died due to VUMC breaching its IACUC regulations in a study involving intubation and ventilator testing. Per the report, the guinea pigs underwent partial anesthesia during the study and died quickly after this procedure. It also states that the IACUC and veterinary staff did not immediately learn of the guinea pig deaths as mandated. VUMC declined to comment further on the nature of the study.
“The PI [Principal Investigator] did not communicate the date, time or nature of the procedures to either the faculty veterinary staff or surgical research training personnel in advance as required by the IACUC in the approved protocol,” the USDA report said. “The guinea pigs in this instance were handled by the PI without the oversight intended by the IACUC.”
After the original four guinea pigs died, the report states that the PI tried to recreate the “unsuccessful” procedure on another guinea pig. A veterinary lab technician who observed the PI’s attempt reported the issue.
“The IACUC conducted a lengthy investigation and determined that the animals experienced undue pain and/or distress during the procedures to test the ventilator,” the USDA report stated.
After the IACUC’s investigation, they discontinued the VUMC procedure nine days later. John Manning, VUMC chief operating officer, self-reported the incident to Axel Wolff, director of the Division of Compliance Oversight at OLAW, per documents obtained by The Hustler. The self-report clarifies that the IACUC suspended the PI from working with live animals at Vanderbilt until further notice.
“The fact that they [the IACUC] revoked his or her [PI] privileges is very good and rare,” Dr. Alka Chandna, People for the Ethical Treatment of Animals vice president of laboratory investigations cases, said.
According to Cole, the USDA generally directs repeat and “indirect” violations to its IES department, which takes action against the facility in violation. These penalties range from warnings to lawsuits to fines, and the IES decides a course of action based on the facility and its violation history, per Cole. However, she also stated the IES can also penalize facilities for an immediate threat against animals, even if the incident has not previously occurred.
“The very fact of referring a case over to the IES should be setting off alarm bells,” Chandna said.
Past incidents
VUMC was fined $7,218.75 by the IES in 2007 after a string of 40 previous violations, according to an 80-page IES report obtained by The Hustler. The report includes 93 exhibits that detail the citations. The violations included using unapproved anesthesia methods, surgeries and ultrasounds, as well as drug usage.
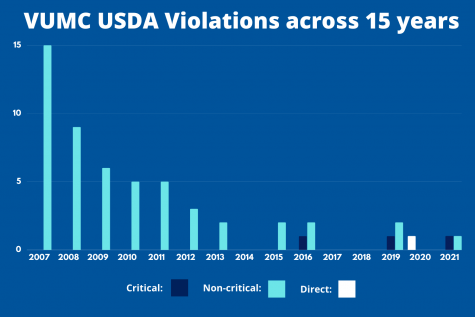
In 2008, VUMC was again fined $8,156 by the IES due to three mishandling citations. according to documents obtained by The Hustler. The first violation involved a galago monkey being killed in a washing machine after a VUMC staff member neglected to thoroughly check nesting boxes for animals before washing them, per the two-page IES report. Another citation detailed how one hamster died and four more had to be later euthanized due to complications with an approved experimental compound. The final violation concerned incomplete IACUC protocol descriptions.
Furthermore, from Dec. 19, 2018, to Oct. 30, 2020, OLAW subjected Vanderbilt to enhanced reporting as a result of repeated mice deaths due to starvation and/or dehydration, per documents obtained by The Hustler. This regimen required Vanderbilt to report new laboratory hires and feeding and watering schedules monthly, instead of only after a regulation breach.
VUMC was cited by the USDA for three more violations in a June 18, 2019, annual inspection. One incident is listed as “critical” and “repeat” and involved a “lack of communication” regarding anesthetics given to gerbils in a VUMC study. Seventeen of the 23 gerbils used in the experiment—which ran from June 2017 to April 2018—died, and their deaths were not reported to the IACUC, according to the inspection report. Chandna referred to critical violations as the “most damning thing” an institution can be issued.
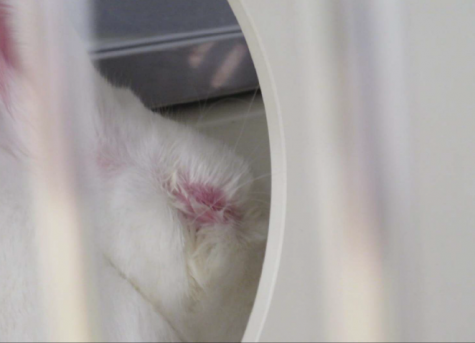
On Feb. 19, 2020, VUMC also received a “direct” non-compliance from the USDA regarding observed abnormalities in six New Zealand white rabbits that, as part of a VUMC study, received drug injections in their eyes on the previous day. Direct non-compliances are issued as a result of adverse conditions that are present at the time of an inspection and critically affect an animal, per the USDA Animal Care Inspection Guide.
According to the report, three rabbits exhibited minor eye swelling and redness, while the other three experienced more extreme inflammation, in addition to lacrimation and forcibly-shut eye(s).
The inspection additionally detailed that the rabbits lacked typical appetites and that some exhibited unusual social feedback, such as being unresponsive to attention.
Per the inspection report, the lab’s protocol said that extreme eye swelling is “very rare.” However, a lab staff member claimed that the rabbits’ symptoms were typical for one to two days after the injections, per the USDA report.
VUMC declined to comment on whether data was published from the studies mentioned in the Feb. 2020 and June 2019 inspection reports.
Research facilities across the country
Per an APHIS database, VUMC has been cited by the USDA for nine total violations—one direct, two critical and six non-critical—from 2015 to 2020, a statistic that is higher than similarly ranked universities and associated medical centers.

Duke University, ranked 12 on the U.S. News Best National University Rankings, accumulated three non-compliances during this time and all were non-critical. Dartmouth College, ranked 13, was issued one critical and three non-critical citations. Brown University, tied with Vanderbilt at rank 14, received two critical and one non-critical violations. No. 16 Rice University received one non-critical violation through these five years. Washington University in St. Louis, tied at No. 16, accumulated two critical citations.
When compared to research facilities that earned similar amounts of research funding from the NIH in 2020, Vanderbilt was issued an above-average number of non-compliances between 2015-2020. The average among five schools with funding similar to that of Vanderbilt was 7.4 violations in comparison to Vanderbilt’s nine. In 2020, Vanderbilt received $377,931,988, placing it at No. 21 in the largest total NIH grants.
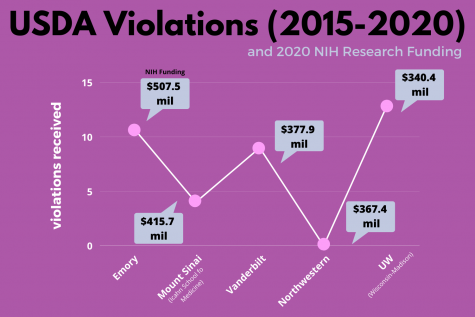
According to the NIH, Emory University was issued eight critical violations and three non-critical violations during the same five-year span while receiving $507,546,965 at No. 18. Icahn School of Medicine at Mount Sinai earned $415,711,940 at No. 20 and amassed four citations, all of which were non-critical. Northwestern University was not issued any non-compliance between 2015 and 2020 but was ranked No. 22 in NIH research grant money, earning $367,414,121. The University of Wisconsin-Madison received 13 citations during this period, six being critical and seven being non-critical. UW is ranked No. 24 and received $338,121,506 in NIH research grants in 2020.
Criticisms of animal experimentation
A VUMC statement backed by the university expressed support for animal research and dedication to the safe conduction of such studies. The statement, sent via email to The Hustler, also referenced previous use of animal research to aid humans.
“Vanderbilt University Medical Center commits considerable manpower and financial resources to its programs in animal research so that the most humane approaches can be used,” the VUMC statement reads.
Chandna stated that Vanderbilt’s animal treatment is “pretty bad” compared to other research facilities. She and Budkie both pushed for Vanderbilt to deviate from animal research.
“Vanderbilt should get out of the dirty, bloody animal experimentation business and redirect its resources toward modern, non-animal research methodologies that will actually help humans,” Chandna’s statement read.
The U.S. Environmental Protection Agency granted $850,000 to Vanderbilt and $848,924 to VUMC on Sept. 10, 2019, to be put toward exploring research methods that limit or eliminate the use of vertebrae animals. Furthermore, as of Feb. 8, 2021, VUMC eliminated the use of live animals in its emergency medicine residency program. This move came after a seven-year campaign by the Physicians Committee for Responsible Medicine.
“Vanderbilt University Medical Center will use the grant to use their Endo Chip technology to research how preexisting diseases affect cell[ul]ar responses to environmental toxicants with a focus on reproductive disorders in women and Vanderbilt University will use the grant to test their organ-on-a-chip to study the blood-brain barrier and potential brain injury after organophosphate exposure,” the EPA press release read.
Chandna advised students interested in research to focus their efforts on methods that have higher success rates than animal experimentation.
“Pursue your dreams in a way that accords with your ethics and look at the data about the failure of animal experimentation and find an advisor who will help you use human-relevant methods,” Chandna said.
Immanual John Milton contributed reporting to this piece.








